 
CO 2 has one carbon atom and two oxygen atoms.Oxygen (O) has an atomic mass of about 16.00 amu.Scientists use the periodic table to quickly refer to information about an element, like atomic mass and chemical symbol. Carbon (C) has an atomic mass of about 12.01 amu. The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights (), compiled as part of the 2021 Table of Standard Atomic Weights 2021. The periodic table of chemical elements, often called the periodic table, organizes all discovered chemical elements in rows (called periods) and columns (called groups) according to increasing atomic number.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound.For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) 28.02 amu. For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. 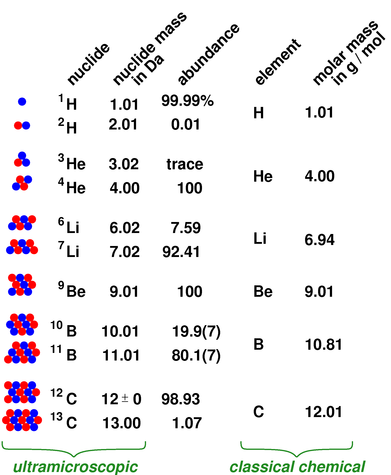
The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). The concept of molar mass can also be applied to compounds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
